Home-based virtual-reality training improves functional recovery in chronic stroke and induces cortical reorganization

Authors: Ioana Stanescu, Oana Vanta
Keywords: chronic stroke rehabilitation, upper limb function recovery, home-based rehabilitation
Community-based and home-based rehabilitation – essential tools in chronic stroke
Can home-based virtual-reality training improves functional recovery in chronic stroke and induces cortical reorganization? Disability is a dramatic consequence of stroke, affecting 50% of survivors, while 30% lose their autonomy and depend on others for daily activities [1]. Rehabilitation is one of the most critical interventions in stroke, and there is evidence that it has benefits in all stroke stages. In the acute stage (first 3 weeks after stroke), rehabilitation is initiated in the stroke unit and continued during hospitalization. In the subacute stage (between 3 weeks and 6 months after the event), stroke severity and predicted outcome would guide the level of care: inpatient rehabilitation facilities or skilled nursing facilities. Despite intensive interventions, 25 to 75% of stroke patients are discharged with significant physical and functional limitations [2].
For more information on stroke, check out:
- Recovery of precise hand movements after stroke
- Motor function after stroke is promoted by early neural network reorganization
To get more insightful perspectives on neurology, watch:
- Interview with Dr. Dana Boering (EFNR Secretary General)
- Interview with Prof. Raad Shakir (Past President WFN)
In chronic stroke stages (after 6 months), rehabilitation training is still beneficial and should be provided in community-based facilities or at home with supervision. There is evidence that physical training is effective in chronic stroke, increasing quality of life and leading to cardiovascular benefits, and should be promoted by community programs. A physically active lifestyle should be encouraged by community and domiciliary interventions. Without constant participation in training programs, a decline in motor and functional acquisition is noticed in stroke patients, and gains obtained by previous inpatient interventions could be lost [2–3].
Robotic devices and virtual reality training in community and home-based rehabilitation
The major challenge for rehabilitation in stroke patients after discharge at home is the successful transition to the community, including an ongoing rehabilitation program. The barriers could be the limited availability of therapists and the distance to the rehabilitation center in the community. This may delay the training schedules and increase patient and caregiver dissatisfaction. One of the latest approaches in stroke rehabilitation is technology-assisted rehabilitation (TAR). TAR has the advantage of providing remote training for patients, adapted to each patient’s needs. Stroke patients could be able to frequently train with minimal therapist supervision and would be provided with constant feedback. TAR includes robotic training systems, virtual reality, and sensor-based training systems.
Previous evidence showed that Virtual Reality (VR) interventions are acceptable and effective in evaluating stroke patients and rehabilitation [4–6]. VR combines devices with specific interfaces with automated training sequences, delivering personalized rehabilitation to patients. One example of VR technology is the Rehabilitation Gaming System (RGS) which is effective in the upper limb rehabilitation of acute and chronic stroke patients [7].
VR training for home-based rehabilitation in stroke patients
A study by Ballester et al. aimed to assess the efficacy and limitations of home VR-based training in stroke patients compared to domiciliary occupational therapy. The authors focused on functional recovery of upper limb motility and cortical network reorganization after treatment [2].
The study, set in Barcelona in 2017, included 39 patients after a first stroke, at more than 12 months from stroke onset (in the late-chronic phase), with mild-to-moderate upper limb (UL) motor deficits (score >2 in proximal UL on the Medical Research Council MRC scale) and without significant cognitive impairment. The patients were familiar with the RGS device previously used during inpatient rehabilitation. Twenty patients were included in the RGS group, and 19 patients were in the control group. The control group received at-home occupational therapy, while the subjects were in the RGS group.
The Rehabilitation Gaming System (RGS) uses an image capture device and allows the patient to control on the screen a virtual body part (like a hand and arm avatar) while performing different tasks. The system is shown in Figure 1.

The tasks and the movements trained by the system are shown in Table 1.

The game parameters are modulated after each task completion and adapted to maintain a performance ratio between 60% and 80% to motivate the patient and provide an optimal challenge for each training session. The RGS system also includes software for the automated evaluation of a patient’s motor performance during training, called AEMF (Automated Evaluation of Motor Function), designed to assess UL motor function without therapist intervention [2]. The AEMF scenario is split into two tasks to evaluate proximal and distal motor function. In the first task, participants had to use planar wiping motions with their arms to remove small cubes from a virtual surface. In task 2, participants were told to flex and stretch their fingers to squeeze a virtual object. These AEMF activities were performed first with the non-paretic limb and subsequently with the paretic limb to ensure that the AEMF tasks were correctly understood. Participants did not receive any direct feedback about their performance. During task execution, the software collected data on hand position and joint rotation (fingers, elbows, and shoulders) to determine the horizontal planar area covered, finger flexion, and finger extension [2].
The study intervention provided subjects with daily sessions of home training with the RGS system, 20-60 minutes/session, 5 days/week, for 3 weeks. Each session consisted of subtasks on the upper limbs’ – hitting, grasping, and placing movements. The control group was performing individual OT at home for the same duration. The expectation was that all participants perform between 1-3 training sessions a day.
At the end of the intervention, all participants could use the gloves and set the VR training system without assistance. Moreover, the authors highlight the significant increase in task difficulty during training, reflecting better performance in both paretic and non-paretic limbs [2].
Benefits of VR training on motor recovery of upper limb function during home-based rehabilitation
In the study conducted by Ballester et al., the authors measured the motor function of the affected upper limb using standard clinical scales [2] presented in Figure 2 below:
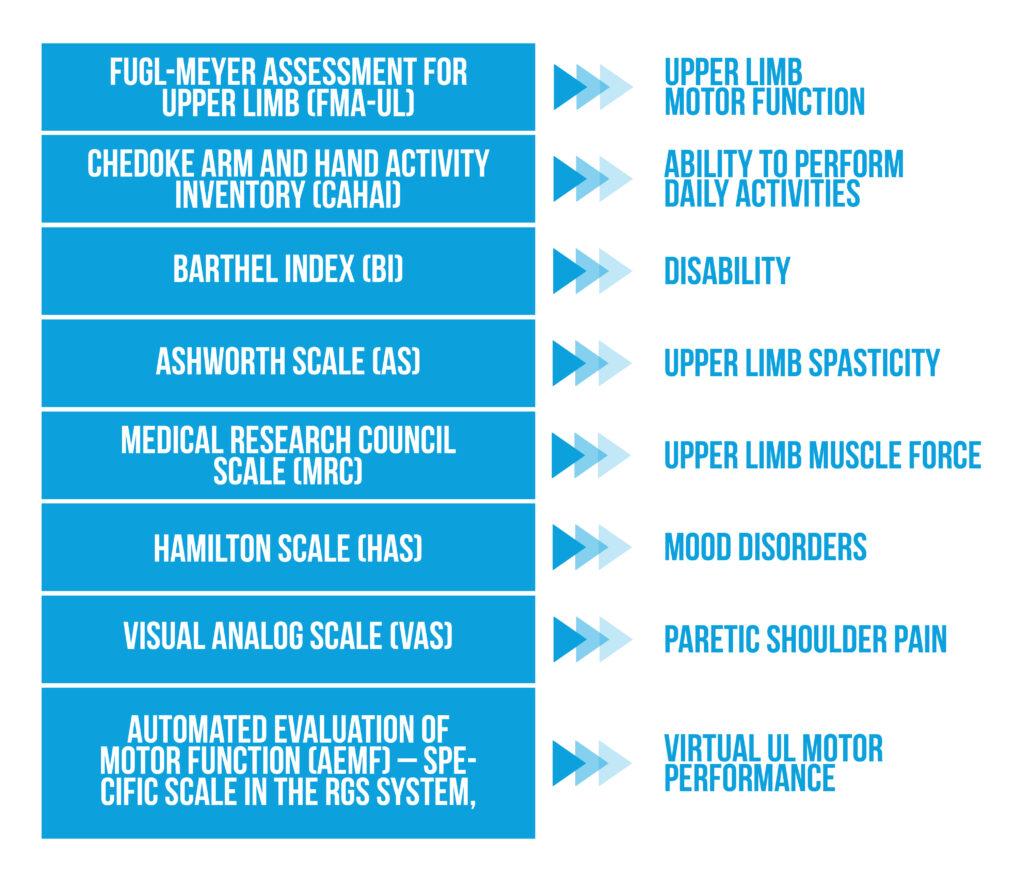
The measurements were performed at inclusion, after 3 weeks of intervention, and at 3-month follow-up. The originality of this study was developing a biomechanical model of the paretic upper limb using captured movements by data gloves and by the screen motion capture device.
No proximal and distal paretic UL motor function improvements were noticed on FMA-UL and MRC scales. Distal upper limb spasticity improved significantly 3 months after VR training (measured on AS). At the end of the intervention, the RGS group showed significantly better upper limb ability in performing daily activities (measured by the CAHAI scale), an effect that was not persistent at follow-up.
The authors pinpoint as an essential finding the detection of functional improvements in the distal part of the paretic upper limb after completing 2 or 3 weeks of VR home training. Repetitive tasks involving reaching and grasping hand movements increase the range of finger flexion and extension. This motor gain in the distal UL function was demonstrated using data collected by the AMEF system, which is another strong point in this study [2].
Looking at these results holistically, the authors noted as a limit to their research, that task difficulty descriptors, AEMF measurements, and NBS all converged, indicating that distal functional improvements were introduced through RGS-based training and were significantly greater for participants in the RGS group when compared to the control group. Furthermore, they also noted that the observed inter-group variabilities were due to the two different interventions applied [2].
VR training induces brain neuroplastic changes in stroke patients
The authors aimed to demonstrate that VR training could induce cortical reorganization in stroke patients. Navigated Brain Stimulation (NBS) procedures were used to demonstrate corticospinal tract integrity and to evaluate the motor map in the primary cortical motor area. The authors used the evaluation of the cortical representation of two hand muscles usually affected by stroke: abductor policis brevis (APB) and extensor-carpi radialis (ECR). The NBS procedure is shown in Figure 3.
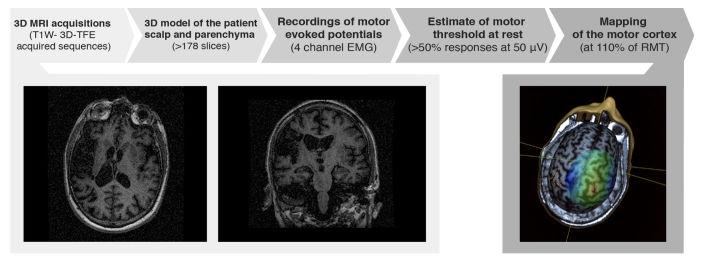
In this study, brain maps of 17 stroke patients were assessed using NBS procedures before and after VR training. The cortical areas representing ABP and ECR muscles were detected on the cortical map. Stimulation efficacy was recorded, and the center of the cortical area which produces MEPs was drawn in both cerebral hemispheres before and after 3 weeks of treatment.
The results showed enhanced excitability in the corticospinal tracts of the affected hemisphere after completing VR training. SE increases in the pathological cortical motor areas after the intervention. Moreover, changes in cortical connectivity were demonstrated by the displacement of the center of the cortical motor area, producing MEPs for both ABP and ECR muscles after the intervention. The authors postulated that these cortical organization changes correlate with the motor gains captured by the clinical scales [2].
Ballister and collab. highlight that the measurement of corticospinal connectivity using the NBS procedure could be used as a biomarker for detecting chronic stroke patients who will respond to rehabilitation treatment [2].
Conclusions
In chronic stroke patients, no motor improvements are expected, and the efficacy of rehabilitation treatments in this phase is still debated. Patients are discharged into the community with limited rehabilitation programs or therapist access. This study conducted by Ballester shows that rehabilitation using new technologies, such as Virtual Reality, is feasible at home without specialist supervision and is accepted by most patients. The Rehabilitation Gaming System (RGS) uses a goal-oriented approach in which the patient tries to control the avatar of its paretic and non-paretic upper limbs while executing complex motor tasks (such as catching moving balls on the screen). RGS parameters are automatically adapted to maintain the game challenge. At the end of the training period, patients were able to perform more difficult tasks, reflecting better performance in both paretic and non-paretic limbs.
This study demonstrates that VR training for 3 weeks induces improvements in motor function of the distal paretic upper limb in chronic stroke patients. Functional gains were obtained for finger flexion and extension movements. These data were obtained using the Automated Evaluation of Motor Function, included in the RGS system. Moreover, VR training decreased spasticity in the distal upper limb. The authors highlight the significant improvements observed in paretic hand abilities during daily activities after 3 weeks of training.
Ballester et al. [2] used brain imaging and neurophysiological methods to demonstrate that VR-based motor training enhanced the excitability of some corticospinal fibers controlling hand function Neuroplastic changes obtained after training reveal that cortical reorganization could be induced in the chronic stage post-stroke by technology-assisted rehabilitation methods, like virtual reality.
Bibliography
- Markus HS. Reducing disability after stroke. International Journal of Stroke. 2022;17(3):249-250. doi:10.1177/17474930221080904
- Ballester BR, Nirme J, Camacho I, Duarte E et al., Domiciliary VR-Based Therapy for Functional Recovery and Cortical Reorganization: Randomized Controlled Trial in Participants at the Chronic Stage Post Stroke. JMIR Serious Games. 2017 Aug 7;5(3):e15. doi: 10.2196/games.6773.
- Winstein CJ, Stein J, Arena R, Bates B et al., American Heart Association Stroke Council, Council on Cardiovascular and Stroke Nursing, Council on Clinical Cardiology, and Council on Quality of Care and Outcomes Research. Guidelines for Adult Stroke Rehabilitation and Recovery: A Guideline for Healthcare Professionals From the American Heart Association/American Stroke Association. Stroke. 2016;47(6):e98-e169. doi: 10.1161/STR.0000000000000098.
- Cameirão MS, Badia SB, Duarte E, Frisoli A, Verschure PF. The combined impact of virtual reality neurorehabilitation and its interfaces on upper extremity functional recovery in patients with chronic stroke. Stroke. 2012 Oct;43(10):2720–8. doi: 10.1161/STROKEAHA.112.653196.
- McEwen D, Taillon-Hobson A, Bilodeau M, Sveistrup H, Finestone H. Virtual reality exercise improves mobility after stroke: an inpatient randomized controlled trial. Stroke. 2014 Jun;45(6):1853–5. doi: 10.1161/STROKEAHA.114.005362
- Rizzo A. Virtual reality and disability: emergence and challenge. Disabil Rehabil. 2009;24(11-12):567–9. doi: 10.1080/09638280110111315
- Ballester BR, Maier M, Duff A, Cameirão M et al., A critical time window for recovery extends beyond one-year post-stroke. J Neurophysiol. 2019;122(1):350-357. doi: 10.1152/jn.00762.2018.










One thought on “Home-based virtual-reality training improves functional recovery in chronic stroke and induces cortical reorganization”